Extended use beyond labelled expiry date for selected lots of Jext® 150 mcg and 300 mcg adrenaline auto-injectors
- 23 November 2023
- Healthcare News
- News
There is currently a shortage of Jext® adrenaline auto-injectors in the UK.

Background to the safety concern
Due to the recent recall of a competitor product, there has been an increased demand for JEXT® in all countries, with demand now far exceeding normal expectations and forecasting. To support and maintain an overall adequate supply, ALK has obtained acceptance from the MHRA to extend the use of specific lot (batch) numbers of Jext® 150 mcg and Jext® 300 mcg auto-injectors, beyond the labelled expiry date by two months. The affected lot numbers are listed in the table below and are also available on www.jext.co.uk.
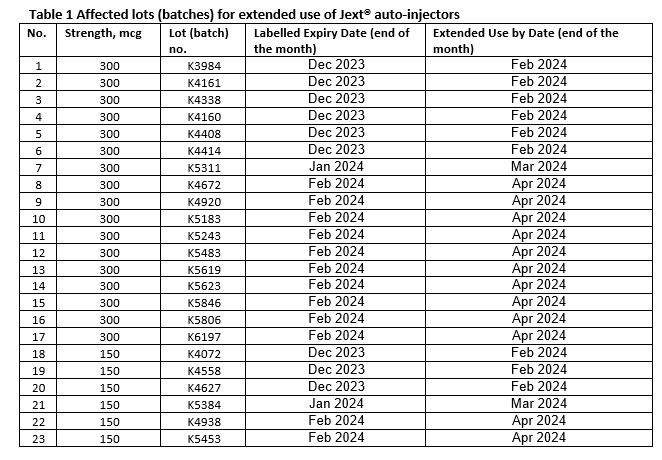
Important: the extended use only applies to the lots of Jext® 150 mcg and Jext® 300 mcg auto-injectors listed above. Patients can continue to use the Jext® auto-injectors of these specified lots safely until the extended use by date as stated above.
This extended use does not apply to any other lot number of Jext® auto-injectors not specified. Patients must continue to adhere to the labelled expiry date on any Jext® auto-injector not covered by the lot numbers above.
Information for patients
• Lot numbers and labelled expiry dates are marked on the end-flap of the box and on the auto-injector label itself.
• The specified lots of Jext® 150 mcg and 300 mcg auto-injectors will continue to work safely over the extended use period beyond the labelled expiry date.
• If you are unsure whether your Jext® auto-injector is affected, talk to your pharmacist.
• The Jext® auto-injectors should continue to be stored as labelled on the pack.
• Towards the end of the extended use period (the end of the month listed in the right column of the table above), a new auto-injector will still need to be obtained.
• You should continue to check periodically the viewing window in the label of your auto-injector to ensure that the liquid inside is clear and colourless. Do not use the auto-injector if the liquid is discoloured.
• You should consult your doctor or pharmacist if you have any concerns regarding this announcement. ALK cannot give specific treatment advice to patients.
Call for reporting
Please continue to report suspected adverse drug reactions (ADRs) to the MHRA through the Yellow Card scheme.
Please report:
- all suspected ADRs that are serious or result in harm. Serious reactions are those that are fatal, life-threatening, disabling or incapacitating, those that cause a congenital abnormality or result in hospitalisation, and those that are considered medically significant for any other reason
- all suspected ADRs associated with new drugs and vaccines identified by the black triangle ▼
You can report via:
- the Yellow Card website https://yellowcard.mhra.gov.uk/
- the free Yellow Card app available from the Apple App Store or Google Play Store
- some clinical IT systems (EMIS/SystmOne/Vision/MiDatabank) for healthcare professionals
Alternatively, you can report a suspected side effect to the Yellow Card scheme by calling 0800 731 6789 for free, Monday to Friday between 9am and 5pm.
When reporting please provide as much information as possible, including information about medical history, any concomitant medication, timing onset, treatment dates, and product brand name.
If you require additional information or have any questions, please contact ALK Customer Services: 0118 903 7940 or infouk@alk.net.




